
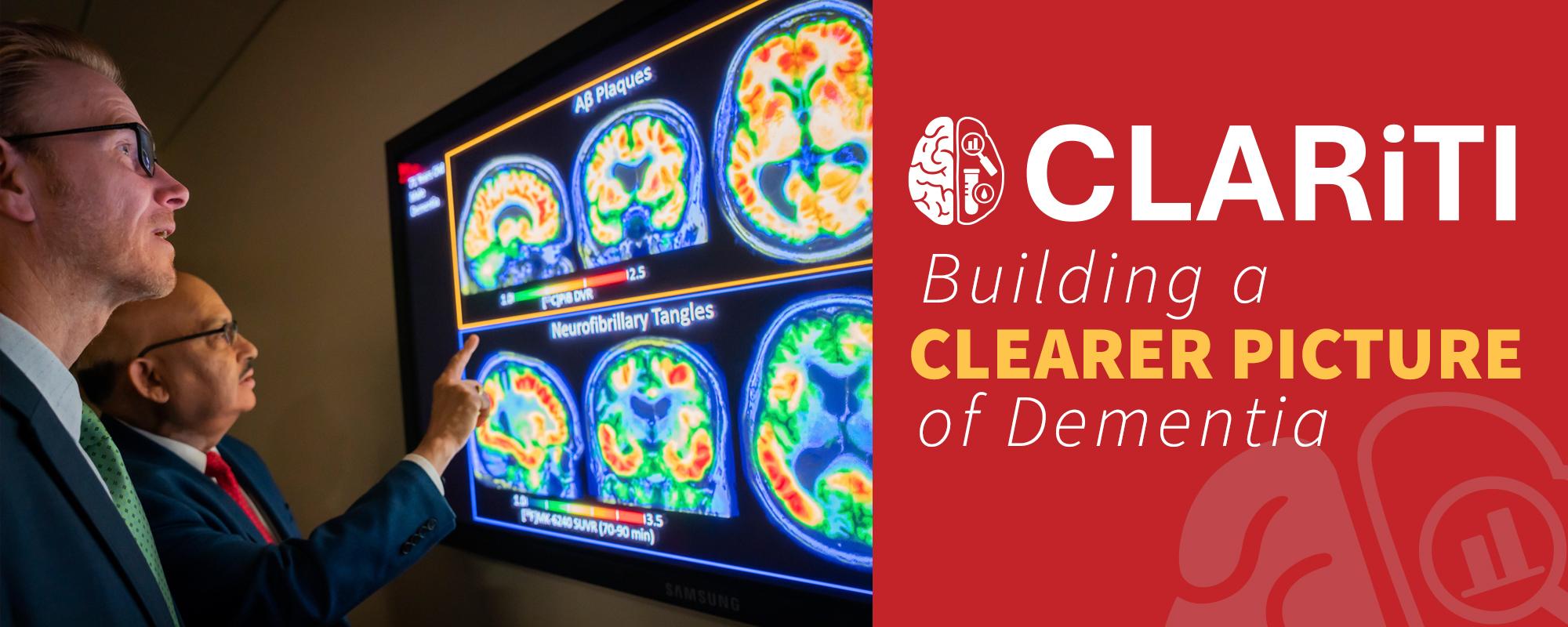
Dementia is rarely simple. For many people living with cognitive decline, more than one underlying brain disease is at work, a reality known as multi-etiology dementia. Yet most large research studies and clinical trials still focus on a single pathology at a time, leaving critical questions about how different types of dementia interact largely unanswered.
The ADRC Consortium for Clarity in ADRD Research Through Imaging (CLARiTI) was built to change that. By bringing standardized PET and MRI imaging into Alzheimer's Disease Research Centers (ADRCs) across the country, CLARiTI is creating the infrastructure needed to study multi-etiology dementia at scale and sharing what it learns broadly with the research community.
Why CLARiTI Matters
ADRCs are uniquely positioned to study multi-etiology dementia. They follow some of the largest and most carefully characterized cohorts of individuals with AD/ADRD, collecting clinical, biomarker, and autopsy data over many years. But until CLARiTI, neuroimaging across these sites has been limited and inconsistent, making it difficult to compare findings or build a unified picture of how mixed pathologies develop and progress.
CLARiTI addresses this by implementing standardized imaging protocols across ADRC sites and integrating the resulting data into the NACC Data Platform, including MRI and PET scans, image analysis results, visual interpretations, neuropathology reports, and plasma biomarker data. Qualified investigators around the world can access this growing resource through the NACC Data Platform for secondary analyses, grant development, and future clinical trials.
With a planned enrollment of 2,000 participants, CLARiTI will create one of the most comprehensive datasets linking standardized imaging, clinical assessments, and pathology findings in the ADRD field. The consortium recently reached an important milestone with its first data release, representing findings from the first 500 enrollments and offering an early look at what this resource will offer the broader research community.
Biomarkers at the Frontier of ADRD Research
A central challenge in studying multi-etiology dementia is detecting and characterizing the multiple pathologies that often co-exist in a single individual. Amyloid and tau imaging have transformed ADRD research over the past two decades, but they capture only part of the biological landscape. CLARiTI is designed to go further, integrating imaging and plasma biomarker data to build a more complete picture of the biological processes driving dementia across diverse populations.
Three researchers contributing to this effort are advancing some of the most promising tools in ADRD biomarker science.

Sara Lorkiewicz, PhD (Stanford ADRC) is working at the intersection of plasma proteomics and Lewy body disease. Many people with Lewy body disease also carry significant Alzheimer's pathology, which affects symptoms, prognosis, and response to treatment. High-sensitivity plasma proteomics offers a scalable, blood-based approach to detecting this co-pathology and identifying disease-specific signatures. Her work supports CLARiTI's Plasma Biomarker Core, which aims to build a foundation for uniform plasma ADRD biomarker characterization across ADRCs, and points toward a future where complex multi-etiology profiles can be identified without relying solely on costly or invasive imaging.

Hartmuth Kolb, PhD (University of Wisconsin) is focused on imaging co-pathologies in neurodegenerative diseases, with particular attention to neuroinflammation, TDP-43, and α-synuclein. While amyloid and tau PET imaging have reshaped how the field understands Alzheimer's disease, they leave much of the biological landscape uncharted. Emerging PET tracers targeting neuroinflammation, α-synuclein, and TDP-43 are opening new possibilities for detecting and characterizing the additional pathologies that drive much of the heterogeneity in dementia outcomes. His work outlines how these tools could be layered into standardized multi-site imaging frameworks like CLARiTI, moving the field closer to a comprehensive, in vivo picture of mixed pathology.

Robert Mach, PhD (University of Pennsylvania, Perelman School of Medicine) is developing [11C]M503, a promising PET tracer designed to image α-synuclein aggregates in vivo. α-synuclein plays a central role in Parkinson's disease, Lewy body dementia, and related conditions, yet the field has lacked a validated imaging tool to detect it directly. Early data on [11C]M503 suggest it may fill this longstanding gap, with selective binding to α-synuclein aggregates and encouraging preclinical PET kinetics. A validated α-synuclein tracer would be a significant advance for the field, enabling more accurate diagnosis of synucleinopathies, better characterization of mixed pathologies, and a potential target-engagement marker for future disease-modifying therapies.
A Shared Resource for the Research Community
CLARiTI is committed to making its data, images, and biofluid-derived values available to qualified investigators. Data and biospecimens collected through CLARiTI are integrated directly into the national databases at NACC and are accessible through the NACC Data Front Door and SCAN/LONI repositories. This commitment to open science is central to CLARiTI's mission: to accelerate discovery by ensuring that the insights gained from standardized, multi-site imaging are available to researchers everywhere.
By fostering collaboration across research centers and building infrastructure that connects imaging, neuropathology, plasma biomarkers, and clinical data, CLARiTI is laying the foundation for a new generation of ADRD research — one that takes the full complexity of dementia seriously and works toward more precise diagnosis, better-designed trials, and ultimately, more effective treatments.
To learn more, visit the CLARiTI website, and register for the 2026 Spring ADRC Meeting, where the CLARiTI team will share the consortium's first data release alongside expert talks on emerging tools and approaches in ADRD biomarker research.
